
As global aquaculture continues to scale, preventing genetic and ecological impacts from escapees has become a priority. One topic attracting scientific and regulatory interest is sterile fish. In this edition of Insights with Ian, we explore the current technologies and their challenges.
There are strong environmental and commercial reasons for using sterile fish in net pen farming. While careful engineering, high maintenance standards, and continuous monitoring can reduce the risk of escapes, they cannot eliminate it entirely. The issue extends beyond the salmon species which are often in the news. European sea bass Dicentrarchus labrax) and gilthead sea bream (Sparus aurata) are the most widely farmed species in the Mediterranean with an estimated 400–500 million individuals per species held in over 20,000 floating cages. Strikingly, the tonnage of wild caught landings (adjusted for fishing effort) shows a strong correlation with the growth of aquaculture and the estimated biomass of escapees entering the wild each year, particularly for sea bream. Freshwater habitats face similar concerns, e.g., Nile tilapia (Oreochromis niloticus) is widely farmed in net pens placed in dams and lakes across Africa, South America and Asia.
Although sterility doesn’t obviate risks associated with the spread of pathogens or inter-species competition, it does provide the ultimate safeguard against interbreeding wild populations.
Commercially, farming sterile fish prevents precocious sexual maturation, which reduces growth and compromises flesh quality. For breeding companies, sterility also helps protect intellectual property and long-term investment in genetic improvement.
So what methods are available for producing sterile animals for aquaculture?
Triploids
The main method currently used is the triploidisation of eggs. Triploidy can result in high rates of sterility and has been widely adopted for rainbow trout farming. However, it is not suitable for many other species. For example, triploid Atlantic salmon are less robust than their diploid counterparts and are therefore not widely used in commercial production.
Gene editing
One method of achieving sterility in diploid fish is through precise gene editing. A decade ago, Norwegian researchers showed that editing the dead end (dnd) gene in 1-cell embryo Atlantic salmon prevented germ cell formation. Later work demonstrated that sterility could be rescued by injecting 1-cell stage embryos with a cocktail of wild type dnd1 messenger RNA (mRNA) and CRISPR-Cas9 constructs targeting dnd. Breeding from rescued dnd crispants paves the way for the large-scale production of sterile salmon.
However, moving this technology from the laboratory to fish farms has proved challenging due to various technical barriers associated with mosaicism and off target edits as discussed in the November 2025 blog. It is also important that the dnd edits are inherited from both parents (i.e. are homozygous for the mutation). The Norwegian Scientific Committee for Food Safety and the Environment (VKM) recently highlighted the risk to wild populations from any fertile heterozygotes present in the gene edited population. They suggested that fish would need to be reared in closed confinement for several generations to prove a stable homozygous genotype.
Given the 4-year lifecycle of Atlantic salmon, it is clear any implementation plan for achieving sterility in this species through gene editing will likely be lengthy and costly.
Gene Silencing
Some of the most promising non-genetic engineering methods of producing sterile diploids involve short RNA molecules and RNA interference (RNAi)
Morpholinos
Morpholinos (MO) are short sequences of RNA with complementary nucleotide bases to the messenger RNA (mRNA) needed to make proteins. When the morpholino binds to mRNA it blocks translation of target genes. If applied sufficiently early in development there is the potential to disrupt the cellular processes that leads to the formation of functional gonads. Solving the problem of getting the morpholino across the thick chorion of the egg without using microinjection is key to scaling the technology for use in aquaculture (Fig. 1).
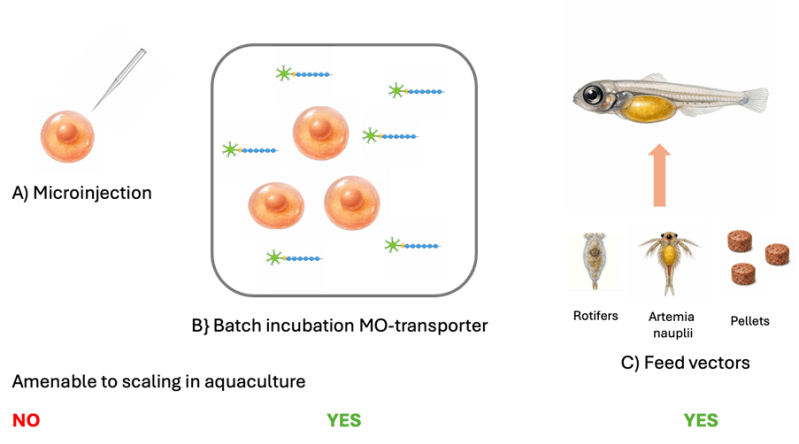
Fig. 1 Delivery methods for introducing siRNAs to cells.
Wong and Zoharin discovered and patented a solution involving the conjugation of a molecular transporter protein called vivo to the dead end morpholino. Experiments with the batch incubation of zebrafish embryos explored the optimal morpholino concentration and application time require to produce 100% sterility. Short incubation times within 5-6h of fertilisation were most successful.
However, only half the fish survived to adulthood with even greater mortality with earlier and longer periods of bath incubation i.e. there was a trade-off between sterility efficiency and survival. The same group recently reported similar experiments with Coho salmon (Oncorhynchus kisutch). Salmon eggs are sensitive to movement once fetilised and therefore treatments were applied before fertilisation. Ovarian fluid was added to the solution in the incubation bath to stabilise the green eggs prior to treatment along with ingredients to prevent water hardening. Temperature, the concentration of dead-end morpholino-vivo conjugate and incubation time all affected survival and sterility rates. Depending on conditions 1-64% of treated embryos survived to the eyed stage and sterility at 14-20 months was less than 10%.
The use of unfertilized eggs is not practical in group spawning fish such as breams, grouper and Seriola species. A recent batch incubation study with sablefish (Anoplopoma fimbria) involving vivo-conjugated dead end morpholino found sterility rates of 8% and 4% respectively in green and fertilised eggs. This suggests the technique might also be applied to batch spawners, but further optimisation and the development of alternative methods for facilitating the uptake of dnd-MO are required (e.g., see here).
CRISPR-Cas13d
A very promising non-GE approach for gene silencing uses the CRISPR-Cas 13d system to edit single-stranded RNA. Since the entire mRNA is a target site for the guide RNA (gRNA) conserved sequences in the dnd1 gene can be identified and used to produce adaptable gRNAs that work across species. In experiments with medaka fish and rainbow trout, fertilised eggs were treated with glutathione to prevent hardening, allowing microinjection of embryos at the 1-cell stage with Cas 13d mRNA and dnd1 gRNA. The procedure achieved 100% sterility and low levels of toxicity with of 80% of injected surviving as in control groups. Microinjection cannot be scaled and is therefore impractical for aquaculture production. Alternative means of delivery will need to be explored before this technology can be considered a practical proposition for aquaculture.
Exploiting natural RNA interference mechanisms
RNA interference or gene silencing occurs naturally in many organisms triggered by double stranded RNA (dsRNA). It likely arose as a defence mechanism against viruses and transposable elements in the genome. Long double stranded RNA is cleaved by RNAase enzymes leading to small interfering RNA (siRNAs) and microRNAs (miRNAs) approximately 21 base pairs long. The pathway that results in the effective and temporary silencing of target genes by short RNAs is explained in Box 1.
Box 1: dsRNA is cleaved by an RNAase enzyme (drosha) to shorter sequences in the nucleus which are then exported to the cytoplasm before binding to another RNase enzyme (dicer) which cleaves it into small interfering RNA (siRNAs) or microRNAs (miRNAs) approximately 21base pairs long. The siRNA is unwound into single sense and antisense stands. The sense strand is cleaved by the protein argonaute 2 (ago2) and discarded whilst the antisense strand is incorporated into the RNA-induced silencing complex (RISC). The RISC assembly then binds to the target mRNA to either inhibit translation or degrade the mRNA effectively silencing the gene (explained in this video).
The first academic paper on RNAi was only published in 1998 and yet the technology proved so impactful that in 2006, it earned two of the authors, Andrew Fire and Craig Mello, the Nobel Prize for Physiology and Medicine. RNAi has been used to systematically study loss of function phenotypes and to develop novel therapies against viral infection, genetic disorders and cancer.
One complication is that siRNAs can function as miRNAs and partial complementarity may result in undesirable miRNA-like off-target effects. Off-target effects need to be investigated and mitigated prior to applying the technology in agriculture and medicine. RNAi-based strategies for the protection and improvement of plant crops that involve genetic engineered dsRNA have been approved by regulators in several countries with commercialisation following (reviewed here). Non-GE applications of RNAi delivered via crop spraying are at an earlier stage of development but have been successful in field trials. Various challenges associated with delivery mechanisms, scalability, off-target effects, risk assessment and regulatory approval are now being actively addressed to expand the technology.

Fig. 2 Korean Rockfish by Open Cage 2. Wikipedia Commons.
In the context of aquaculture, a recent study involving Korean rockfish Sebastes schegelii tested a non-invasive method of delivering dsDNA through feeding using larvae rotifers, artemia nauplii and commercial pellet food as vectors to deliver an Escherichia coli strain engineered to express dsRNA targeting the dnd1 gene (Fig. 1). Around 50% of fish become sterile, whilst lower than with microinjection, this represents a good basis for further optimisation to achieve higher rates.
The use of fluorescent reporter molecules to confirm the delivery of RNAs to their target in combination with subsequent egg sorting would be one way to achieve the desired 100% sterility rate.
Conclusions
There is a compelling environmental and commercial rationale for deploying sterile diploid fish in net pen farming. Unfortunately, for now the most effective technologies remain in the research and development phase. Among them, gene silencing approaches appear particularly promising and with sufficient research investment and regulatory approval, could well become mainstream within 5-10 years.
The winning solution will undoubtedly be the one that delivers 100% sterility, high animal welfare standards and has the lowest implementation costs. Other environmental objections to net pen farming, namely competition and disease risks, can also be mitigated. Lower stocking densities and genetic selection for greater disease resistance, would help reduce the spread of pathogens from farmed to wild fish. Furthermore, over the longer term, ongoing domestication is expected to reduce the survival and competitiveness of escapees.







